- Call Us: +91 9686433300
- E-Mail : contactus@qualitcert.com
- Call Us: +91 9686433300
- E-Mail : contactus@qualitcert.com
-ISO Certification-
QUALIT
CERT
ISO 13485 Certification & Consulting Service in Saudi Arabia
Qualitcert provides comprehensive ISO 13485 certification and consulting services in Saudi Arabia, supporting medical device manufacturers in establishing and maintaining quality management systems (QMS) that comply with international standards and regulatory requirements. With a team of experienced consultants and auditors specialized in the medical device industry, Qualitcert assists organizations in navigating the complexities of ISO 13485 certification, from initial gap analysis to implementation, training, documentation, and audit preparation. By leveraging industry best practices and a risk-based approach, Qualitcert helps clients optimize their QMS to enhance product quality, safety, and regulatory compliance, ensuring the reliability and effectiveness of their medical devices. Through tailored consulting solutions and practical guidance, Qualitcert empowers organizations to streamline their processes, minimize risks, and achieve ISO 13485 certification, enabling them to access global markets and demonstrate their commitment to delivering safe and effective medical devices in Saudi Arabia.
Please Reach Us Today

Please Reach Us Today


Approach and Methodology used to implement Management System Standard
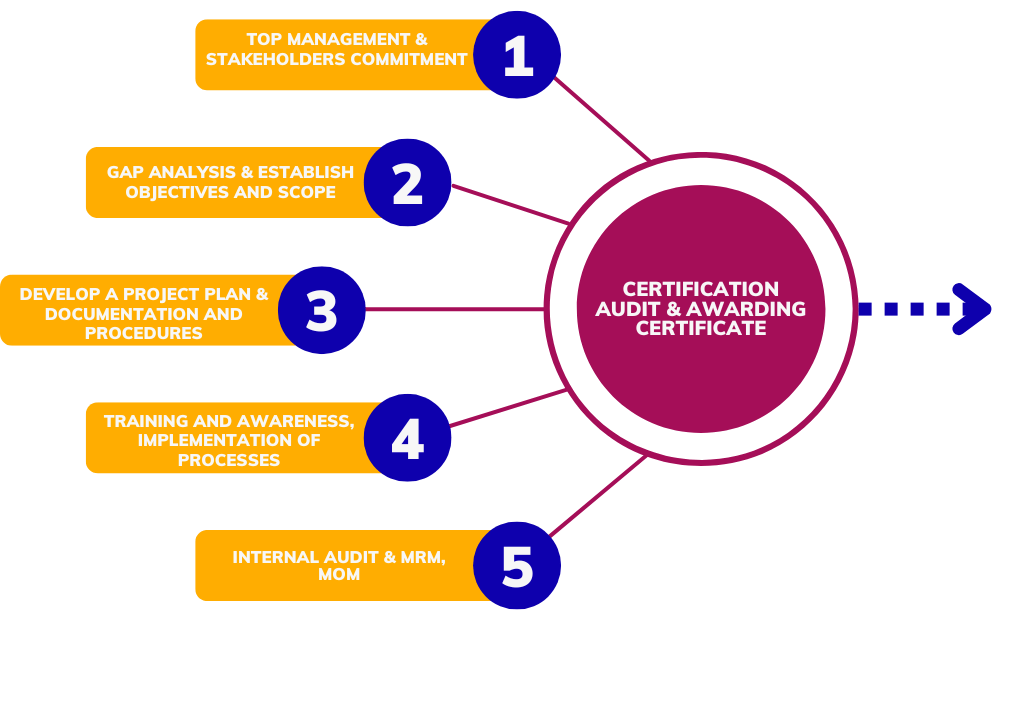
Implementing an ISO standards involves a structured methodology to ensure that the organization effectively meets the requirements of the chosen standard and achieves certification. Sometimes defined methodology may vary depending on factors such as the size of the organization, its industry, and the complexity of the ISO standard being implemented, the following steps provide a basic framework


OUR
Process
1, Determine the ISO Standard
2. Understand the Requirements
3. Training and Awareness
4. Implement the System
5. Internal Audit
6. Certification

Benefits of having ISO Certification
Enhanced Credibility and Reputation
Legal and Regulatory Compliance
Enhanced Customer Satisfaction
Access to Global Markets
Environmental Sustainability
Information Security
Our Achievements and Success
Professional Experts
0
+
Years Experience
0
+
Projects
0
+
Satisfied Customers
0
%
Our Clients





OUR
SERVICES
ISO 13485:2016
ISO 27001:2022
ISO 20000-1:2018
ISO 29001
Our presence in Bangalore
Re-Design your system, create raving fan clients. Transform your business , Go Global
- ISO Certification in Oragadam Chennai
ISO Certification in West Chennai
- ISO Certification in North Chennai
- ISO Certification in Tambaram Chennai
- ISO Certification in Pallavaram Chennai
- ISO Certification in Sholinganallur Chennai
- ISO Certification in Adayar Chennai
- ISO Certification in Velachery Chennai
- ISO Certification in Taramani Chennai

